 Cleanzine: your weekly cleaning and hygiene industry newsletter 17th July 2025 Issue no. 1171
Cleanzine: your weekly cleaning and hygiene industry newsletter 17th July 2025 Issue no. 1171
Your industry news - first
The original and best - for over 20 years!
We strongly recommend viewing Cleanzine full size in your web browser. Click our masthead above to visit our website version.
Just how much do you know about cleaning with bleach?
Rafael Cobos AMIMarEST, of FutureClean Systems, writes...
Recent work with a number of different (and unconnected) clients has revealed problems caused by a similar source - Bleach; Sodium Hypochlorite to be more exact. In one example, bleach was used to disinfect a large tank of water, initially through super chlorination and then to maintain clean water. Another example, which is more worrying, is the use of bleach for routine janitorial cleaning.
These are just a couple of many examples we've seen, particularly in cleaning applications which is also a recurring theme amongst smaller cleaning operations.
Warning: article contains graphic image
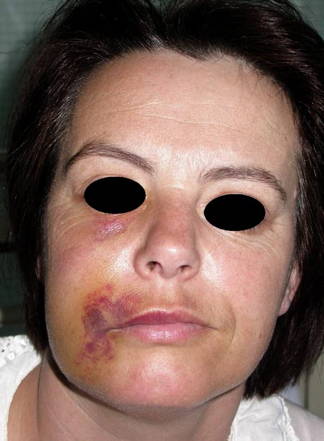 Chlorine (in the form of a Hypochlorite ion) is a widely used disinfectant in the water industry and is highly effective against a wide range of bacteria and viruses. So, it didn't come as any great surprise to see this form of disinfection. What was a surprise was the complete lack of awareness about the risks of using and handling Hypochlorite-based products and in particular the use of bleach for cleaning. Because of the lack of awareness of the risks, Hypochlorite-based products were handled without any due regard for safety to the users themselves, their nearest neighbours or the surfaces the product is applied to.
Chlorine (in the form of a Hypochlorite ion) is a widely used disinfectant in the water industry and is highly effective against a wide range of bacteria and viruses. So, it didn't come as any great surprise to see this form of disinfection. What was a surprise was the complete lack of awareness about the risks of using and handling Hypochlorite-based products and in particular the use of bleach for cleaning. Because of the lack of awareness of the risks, Hypochlorite-based products were handled without any due regard for safety to the users themselves, their nearest neighbours or the surfaces the product is applied to.
Depending where in the world different legislative bodies regulate the use and handling of chemicals in the workplace, they usually require a minimum standard of protection for the skin and the face. In the UK and the US, the Health & Safety Executive and the Occupational Safety & Health Administration respectively, regulate the safety of the workplace and mandate the wearing of personal protective equipment (PPE - e.g. face protection and suitable gloves); they also require a safe working environment through adequate ventilation, as well as emergency measures in place should an accident happen. Little did those involved in the examples above know that all these safety measures are for the use and handling of bleach in the workplace.
Bleach is a powerful oxidising agent and as such is corrosive to substances and surfaces. Just like Sodium Hydroxide, bleach causes severe burns to skin, eyes - and in a vapour form, will cause severe and acute burns to the lungs and mucous membranes. One of the major health hazards is the liberation of toxic Chlorine gas when bleach is mixed with other cleaning products, acids and ammonia products being the principle causes. As a salient reminder, Chlorine gas was used as a chemical warfare agent in World War 1.
When bleach is added to ammonia cleaning products that have become mixed with urine, highly toxic Chloramines are produced which can result in similar health problems as those caused by Chlorine gas.
Generally, the mixing of bleach with other cleaning products is purely unintentional. For example, it is quite common for a toilet descaling product to contain acids - Acids dissolve water scale - and after descaling, bleach is introduced to the bowl without proper regard to the thorough rinsing off of the acidic product first. Chlorine gas is immediately released just as you are bending over the bowl.
Very severe cases of inhalation of chlorine gas can cause pulmonary edema; if ingested, acute - leading to chronic - poisoning cases have been noted.
One of the most common areas in which bleach is used in the home is the kitchen; more specifically worktops and sinks. These worktops, including stainless steel skins and worktops, are highly sensitive to bleach. The chloride ion (Cl-) penetrates the protective surface film (stainless steel 316) and reacts to cause corrosion. This corrosive reaction causes microscopic cracks that, when the steel is subjected to mechanical stress, propagate - leading to sudden failure. On laminated worktops, like Formica, bleach oxidises the polymers and bleaches the colouring. This slowly breaks down the material as the polymers break, making the surface more brittle and causing cracks to appear. You may have noticed that accidental splashes of bleach, if left on the surface, leave an indelible mark.
As we have already seen, Chlorine is a widely used disinfectant in the water industry and is highly effective against a wide range of bacteria and viruses. We have already seen the dangers of uncontrolled use of chlorine (in bleach), but in a controlled environment, chlorination is highly cost-effective and relatively safe. The one major problem with chlorination is the presence of organic matter, since chlorine reacts to produce potentially carcinogenic trihalomethanes.
From a cleaning point of view, the problems and risks of using bleach as a disinfectant completely dwarf the biocidial properties. There are many non-bleach based products on the market, one good alternative is using Quaternary ammonium compounds (Quats), benzalkonium chloride being just one such example. However, they are not quite as effective in killing viruses and bacteria, so longer contact times are needed, but they don't have potentially dangerous reactions that bleach could cause. The market for safer disinfectants is ever evolving and new biocides are showing much better efficacy and wider kill capability.
Image courtesy of www.nature.com shows some of the damage careless use can cause
Rafael Cobos AMIMarEST
T: (01946) 395 548
E: [email protected]
W: www.futurecleansystems.com
6th November 2014







