 Cleanzine: your weekly cleaning and hygiene industry newsletter 26th June 2025 Issue no. 1168
Cleanzine: your weekly cleaning and hygiene industry newsletter 26th June 2025 Issue no. 1168
Your industry news - first
The original and best - for over 20 years!
We strongly recommend viewing Cleanzine full size in your web browser. Click our masthead above to visit our website version.
Hospital-grade microfibre said to eliminate risk of recontamination
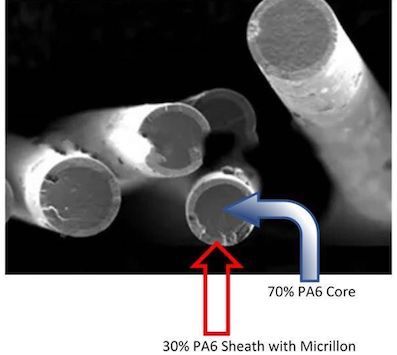 UMF Corporation says its Micrillon microfibre is setting a new standard as the first hospital-grade microfibre that can be converted into a wide range of long-lasting, reusable products, including towels; cubicle curtains; socks; gloves; walk-off mats; reusable N95 rated face masks, and colour-coded microfibre cleaning products.
UMF Corporation says its Micrillon microfibre is setting a new standard as the first hospital-grade microfibre that can be converted into a wide range of long-lasting, reusable products, including towels; cubicle curtains; socks; gloves; walk-off mats; reusable N95 rated face masks, and colour-coded microfibre cleaning products.
Leading manufacturers are using these high-performance microfibre yarns to develop a new standard for materials targeting infection prevention in hospitals, hotels, cruise lines, long term care, and many other industries.
"The introduction of Micrillon microfibre puts an end, once and for all, to any question about recontamination related to reusable products," says UMF Corporation CEO George Clarke. "Some manufacturers and distributors of disposable products - including cubicle curtains, microfibre mops and wipers - have generated controversy around the risk of reusable products, such as microfibre wipers, recontaminating a patient room, even after being laundered. This illusive thinking ignores the fact that after laundering, wipers used in hospitals are immersed in an EPA-registered disinfectant before use - effectively killing bacteria and inactivating viruses."
Micrillon is a rechargeable polymer additive that can be incorporated into manmade fibres, films and plastics and then charged with chlorine molecules. The chemistry recharges for the life of the product and will not leach into the environment. When microbes come into contact with a Micrillon surface, they are eliminated; viruses are inactivated.
UMF's series of high-performance Micrillon yarns, including sheath and core, bicomponent segmented pie, and hollow core segmented pie, demonstrate significant antiviral properties against Human Coronavirus, which causes Covid-19, and Human Influenza A H1N1 virus in just minutes. Micrillon also demonstrates 100% antibacterial elimination of Staphylococcus aureus (MRSA) and E. coli 0157:H7.
"UMF has dedicated significant time and resources developing the unrivaled Micrillon cleaning system," adds George Clarke. "Micrillon products can physically remove everything from a surface and absorb it into the textile, where the disinfectant dwell time exceeds that required to inactivate and kill microbes. These durable, reusable products are cost effective and sustainable. They significantly reduce the burden on the medical products waste stream."
Dr Mina Izadjoo, president and chief science officer at contract services organisation Integrated Pharma Services, which conducted Micrillon's antimicrobial efficacy testing, reports: "We had the opportunity to test Micrillon technology and our results demonstrated significant antimicrobial activity against various pathogens. We are proud that our test and evaluation may lead to the advancement of much-needed infection control measures against hard-to-treat and drug-resistant pathogens."
UMF also recently introduced Klorese, a private labeled disinfectant, now EPA-registered in 50 states of the US. Klorese has broad spectrum effectiveness against Human Coronavirus, C. diff spores, Candida auris, MRSA and many additional bacteria, fungi, spores, viruses and mycobacteria. Together, Micrillon true colour-coded microfibre products and Klorese offer hospital-grade, facility-wide, clinically acceptable cleaning and disinfection.
UMF is a member of the American Reusable Textile Association (ARTA) microfibre Committee, which promotes the benefits and performance capabilities of reusable microfibre products and refutes the misinformation surrounding reusable products.
7th October 2021







